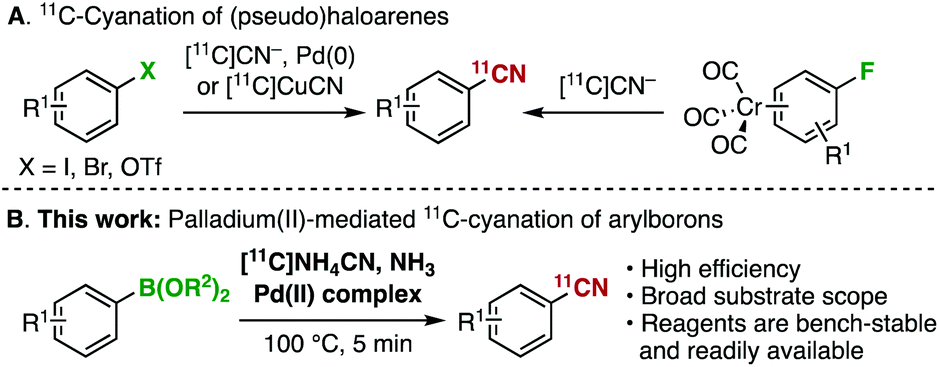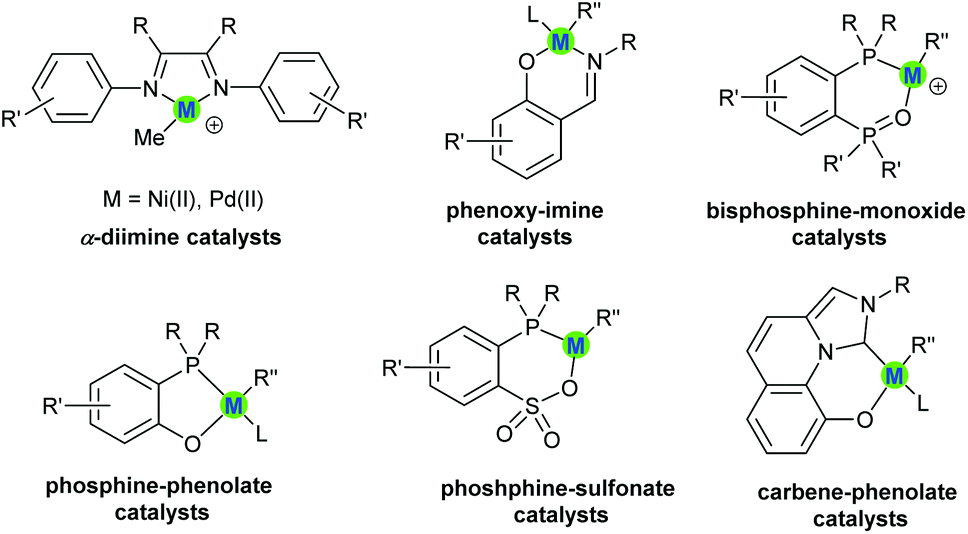
Custom-made polar monomers utilized in nickel and palladium promoted olefin copolymerization - Polymer Chemistry (RSC Publishing) DOI:10.1039/D1PY00492A
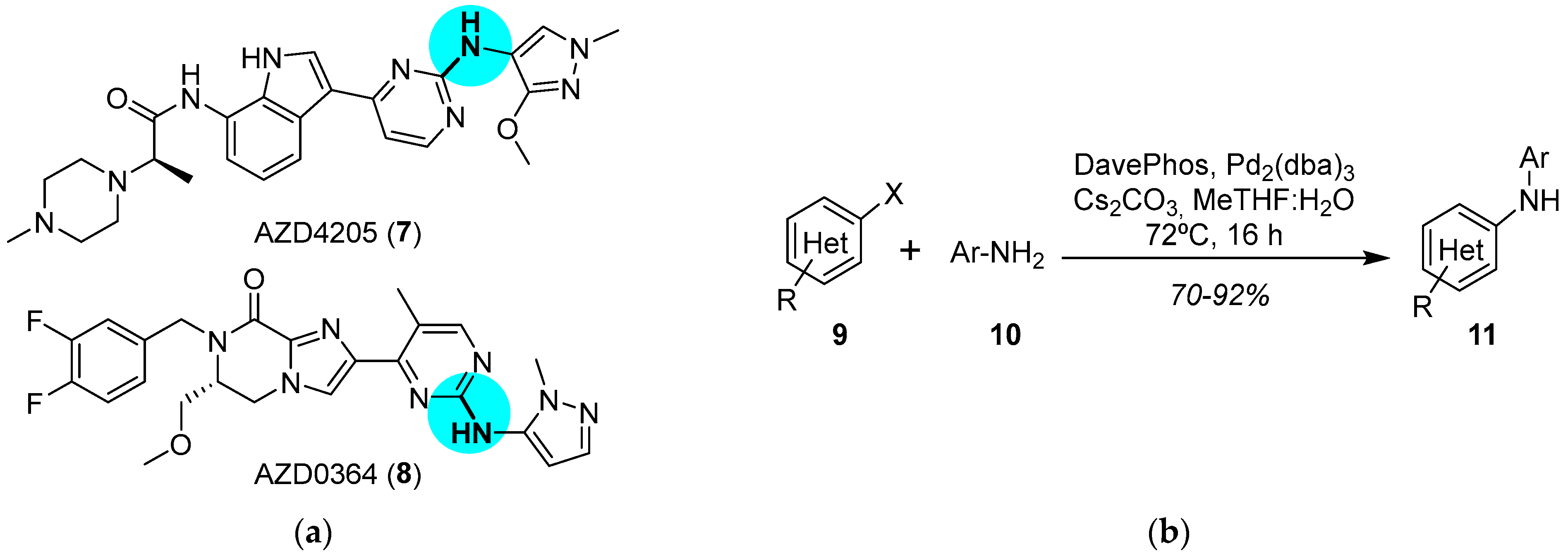
Catalysts | Free Full-Text | Iridium- and Palladium-Based Catalysts in the Pharmaceutical Industry | HTML
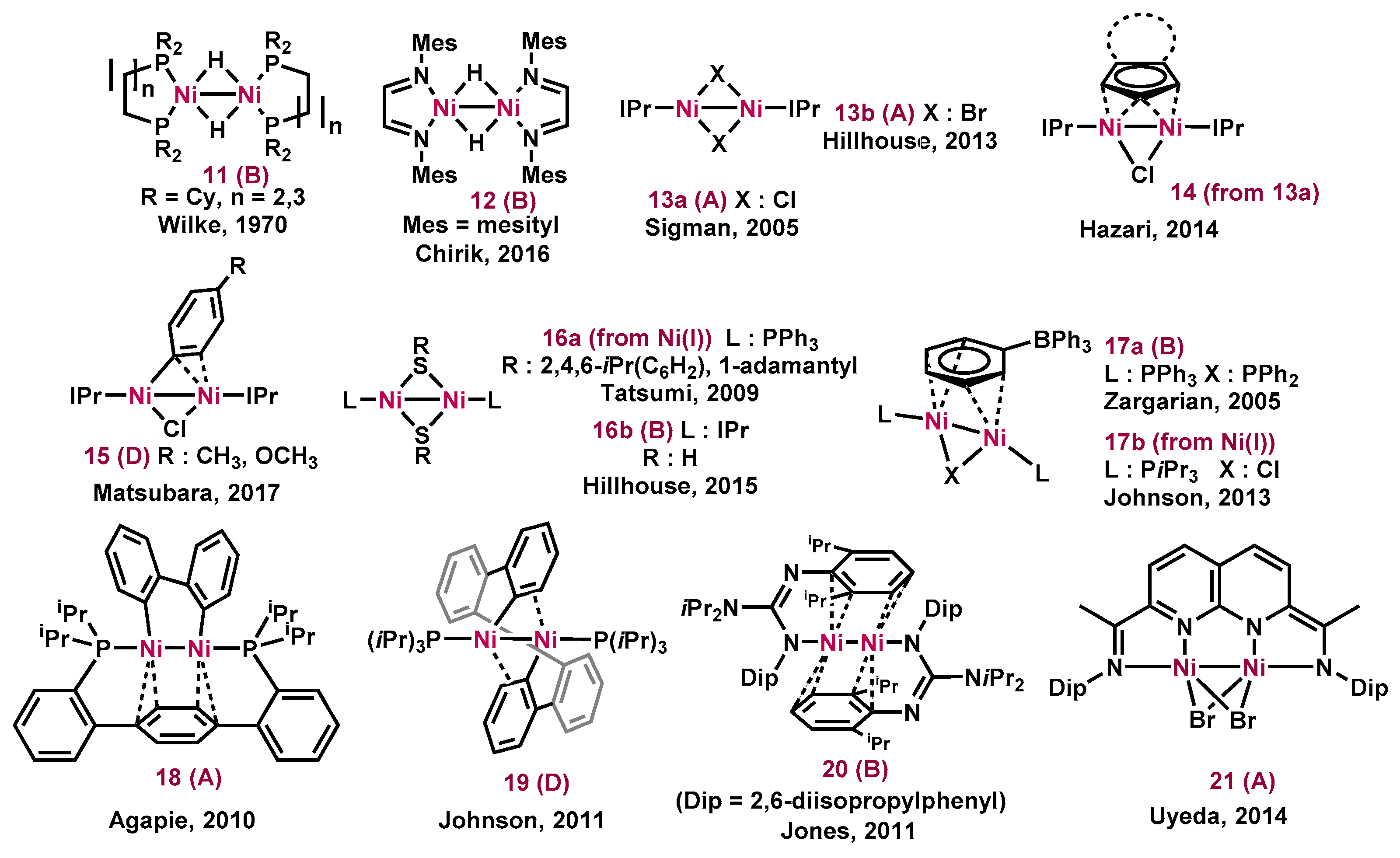
Molecules | Free Full-Text | Dinuclear Nickel(I) and Palladium(I) Complexes for Highly Active Transformations of Organic Compounds | HTML

Evaluating differences in the active-site electronics of supported Au nanoparticle catalysts using Hammett and DFT studies | Nature Chemistry

Photoinduction of palladium single atoms supported on defect-containing γ-AlOOH nanoleaf for efficient trans-stilbene epoxidation - ScienceDirect

Oligonucleotide Bioconjugation with Bifunctional Palladium Reagents - Jbara - 2021 - Angewandte Chemie International Edition - Wiley Online Library

Evolution of palladium sulfide phases during thermal treatments and consequences for acetylene hydrogenation - ScienceDirect
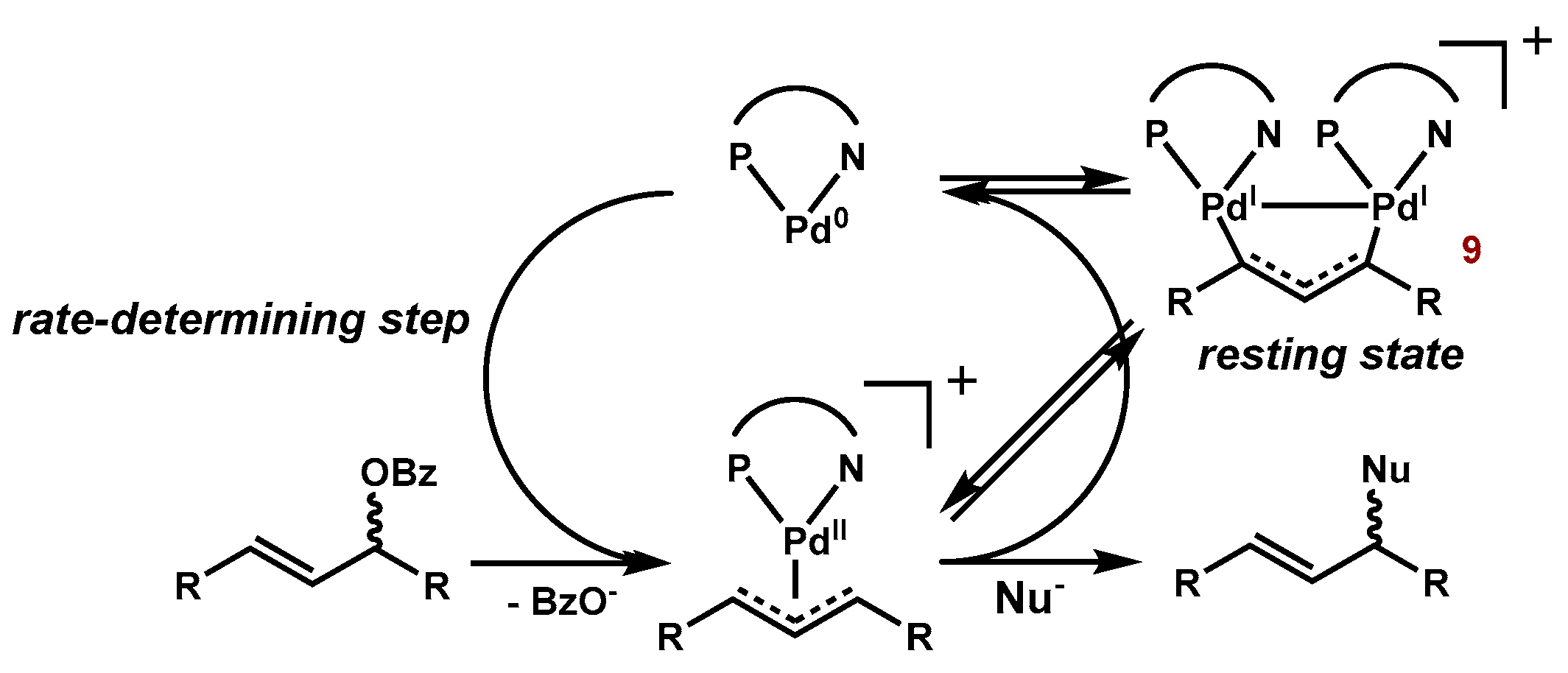
Molecules | Free Full-Text | Dinuclear Nickel(I) and Palladium(I) Complexes for Highly Active Transformations of Organic Compounds | HTML

PDF) Boron Nitride as a Novel Support for Highly Stable Palladium Nanocatalysts by Atomic Layer Deposition

PDF) Synthesis and Characterization of PVP-Stabilized Palladium Nanoparticles by XRD, SAXS, SP-ICP-MS, and SEM
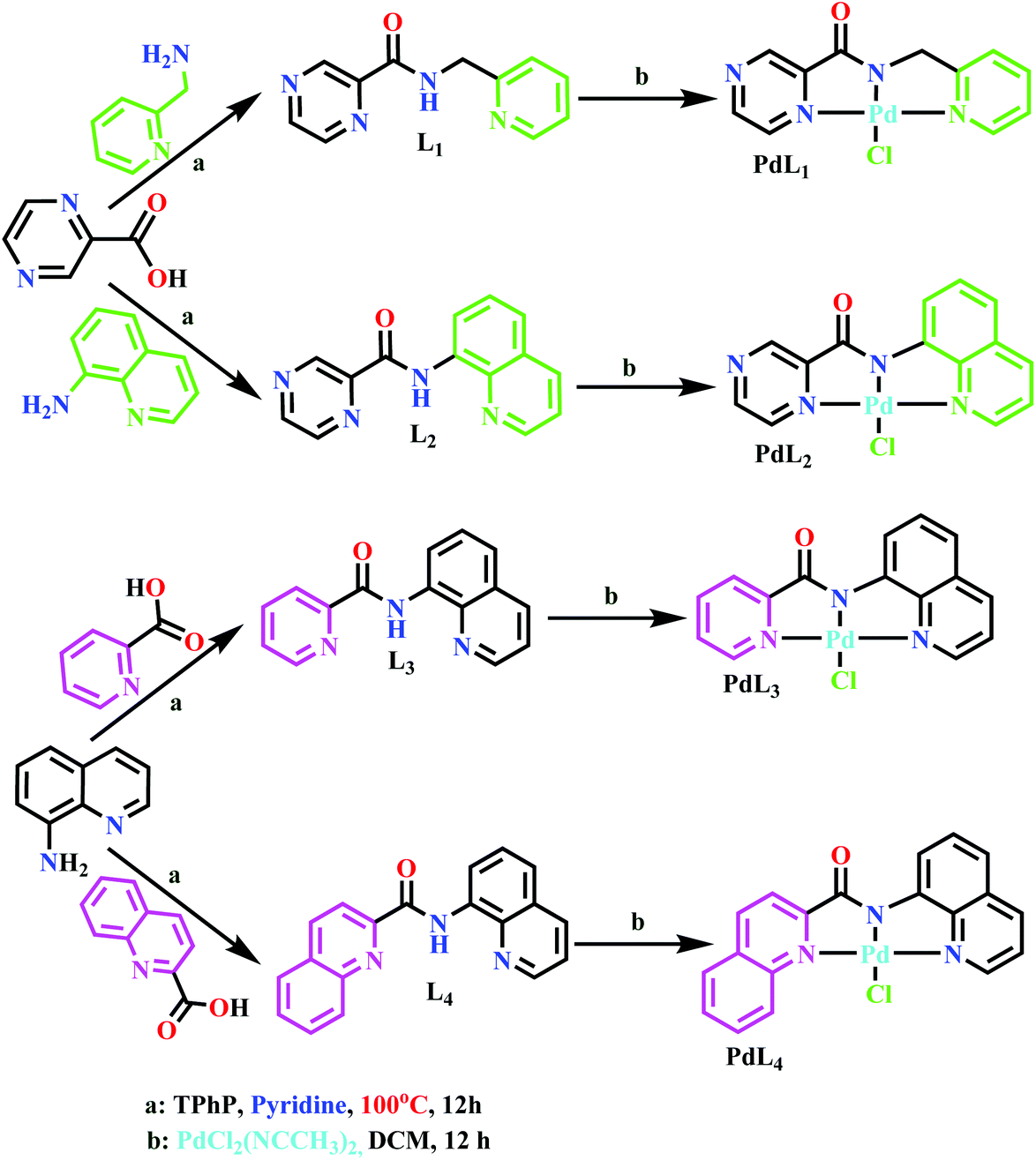
Role of π-conjugation on the coordination behaviour, substitution kinetics, DNA/BSA interactions, and in vitro cytotoxicity of carboxamide palladium(ii) complexes - Dalton Transactions (RSC Publishing)

Synthesis, characterisation, crystal structures and biological studies of palladium( ii ) complexes containing 5-(2-hydroxy-3-methoxy-phenyl)-2,4-dihy ... - New Journal of Chemistry (RSC Publishing) DOI:10.1039/C8NJ03714K
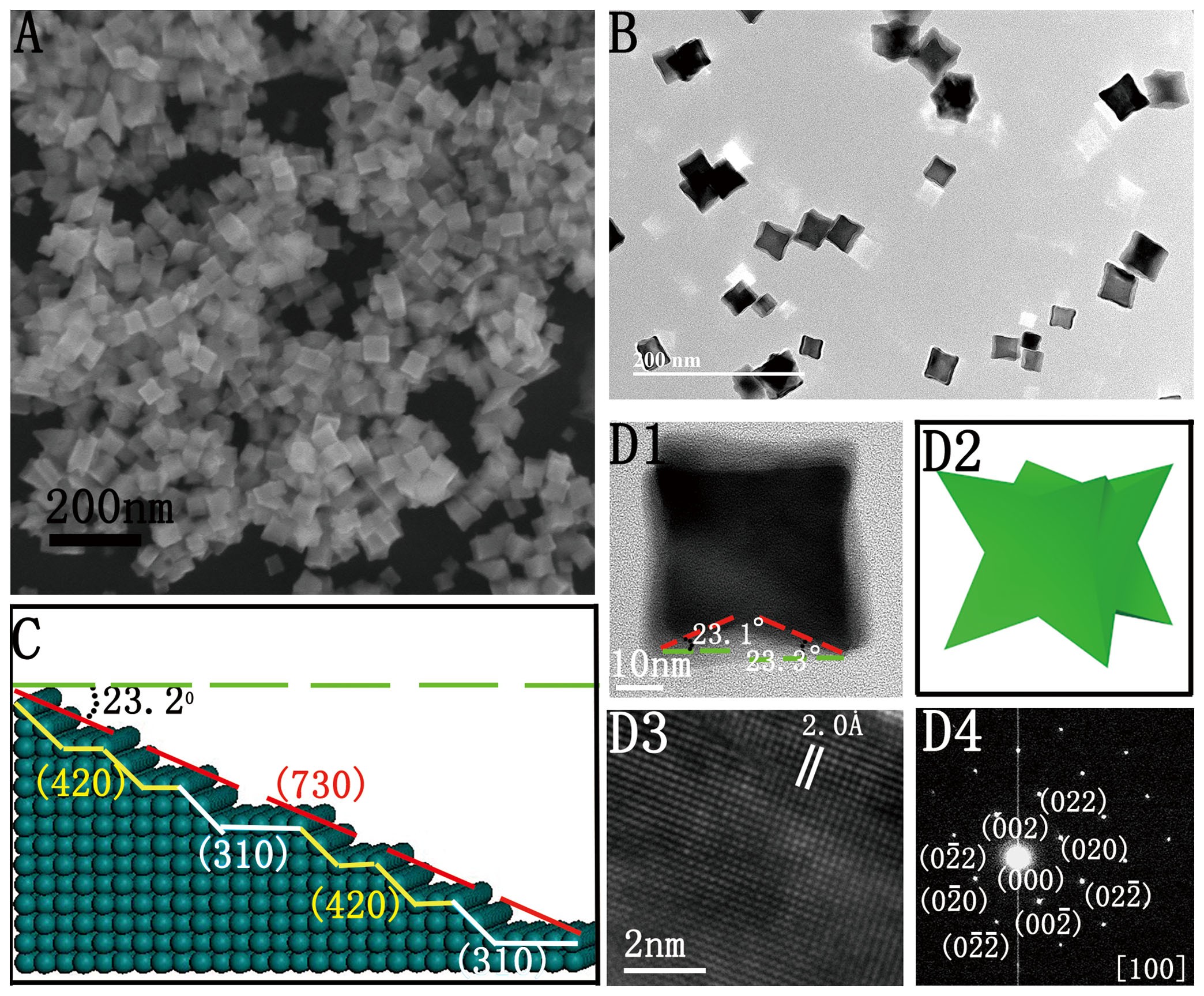
Large-Scale Synthesis of Palladium Concave Nanocubes with High-Index Facets for Sustainable Enhanced Catalytic Performance | Scientific Reports
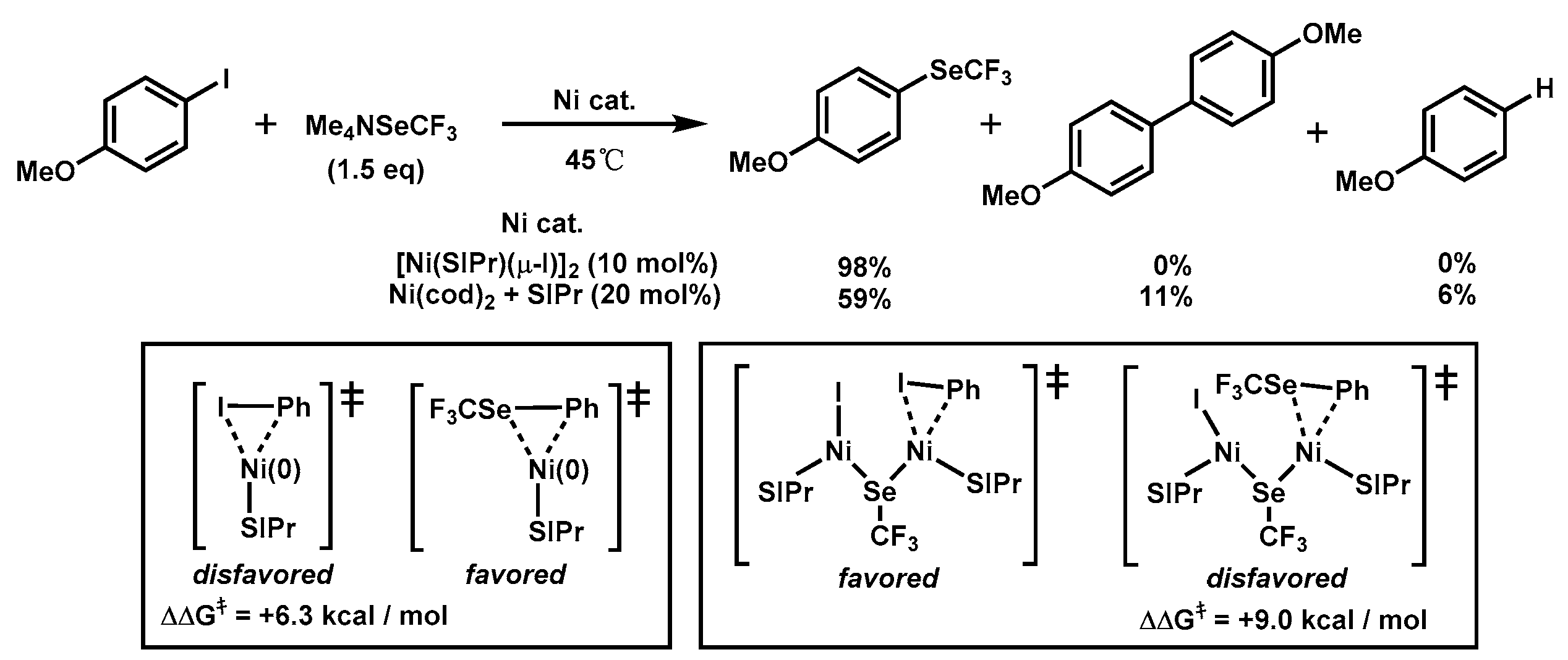
Molecules | Free Full-Text | Dinuclear Nickel(I) and Palladium(I) Complexes for Highly Active Transformations of Organic Compounds | HTML

Three-dimensional atomic mapping of ligands on palladium nanoparticles by atom probe tomography | Nature Communications